Projects
We welcome industrial collaborations from around the globe. Please contact Dr.Georgios Nikiforids and Mr.Haotian MA for more information.
Featured
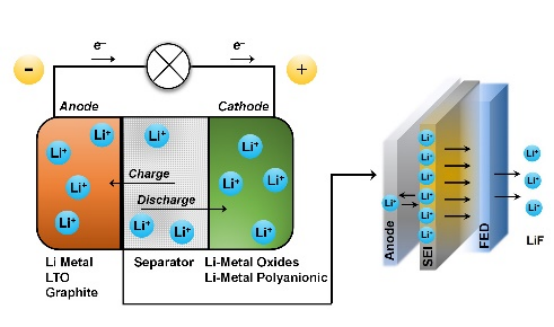
The use of fluorinated ether derivatives (FED), such as 1,1,2,2-Tetrafluoroethyl 2,2,2-trifluoroethyl ether, has shown potential for improving the solid electrolyte interface (SEI) layer in lithium-ion batteries. The table below outlines the projected cell characteristics with different electrode materials, focusing on enhanced SEI stability, cycle life, energy density, and electrolyte compatibility across various lithium-based materials, including LTO (Li₄Ti₅O₁₂), Li-Metal Oxides (LiCoO₂, LiMn₂O₄, LiNi₁/₃Co₁/₃Mn₁/₃O), and Li-Metal Polyanionic compounds (LiFePO₄, LiMnPO₄).
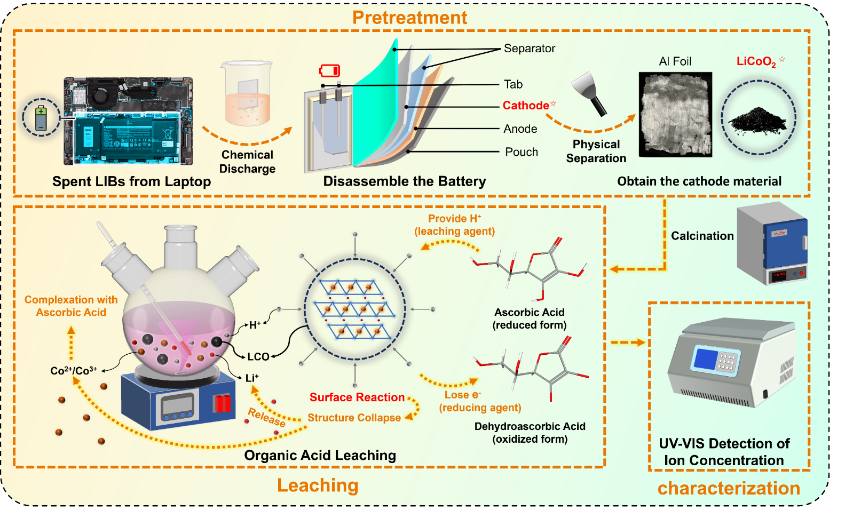
The proposed recycling process introduces organic acids during the leaching phase to extract high-value metals such as graphite, cobalt, lithium, and copper—identified as critical raw materials (CRMs). Organic acids like oxalate, citric acid, malic acid, and aspartic acid, combined with oxidant agents, offer a greener solution due to their low environmental impact. These acids are biodegradable, produce minimal hazardous waste, and allow selective leaching, making them a sustainable choice for metal extraction from lithium cobalt oxide (LiCoO₂) consumer batteries.
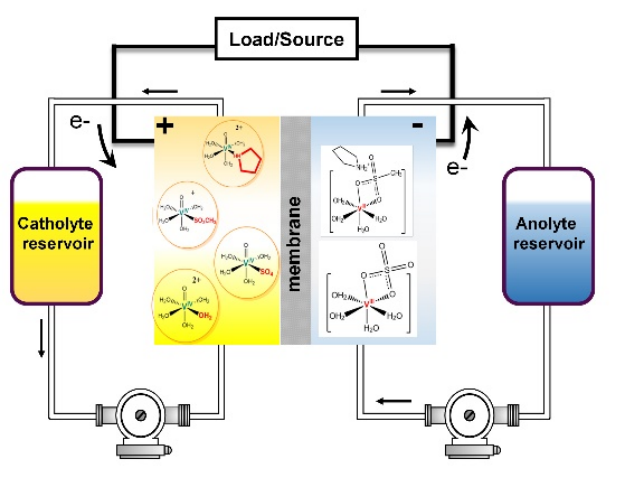
A Vanadium Redox Flow Battery (VRFB) utilizing pyrrolidine and methanesulfonic acid (e.g., PyrrH⁺CH₃SO₃⁻) combined with H₂O at varying mass ratios is proposed as a more functional electrolyte alternative. The formation of highly soluble V⁴⁺ and V³⁺ complexes is attributed to the deprotonation of the displaced pyrrolidinium cation and complexation of the vanadyl cations by the amine ligand. This process enhances the energy density of the redox flow battery by a factor of 2, without the need for additional additives.
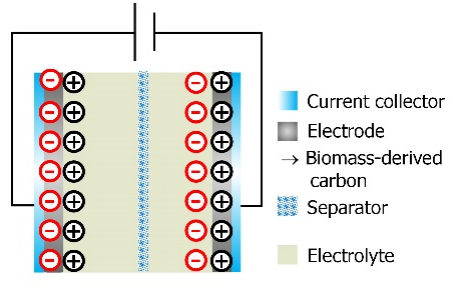
Supercapacitors (SC) utilizing protic ionic liquids (PIL) and deep eutectic solvents (DES) in both liquid and gel-like forms. The diagram illustrates the mechanism for ionic mobility within an aqueous gel based on polyvinyl alcohol and formamide, combined with a mixture of two nitrate salts, lithium nitrate and pyrrolidinium nitrate. This innovative composition enhances ionic conductivity and energy storage capabilities in supercapacitor systems.
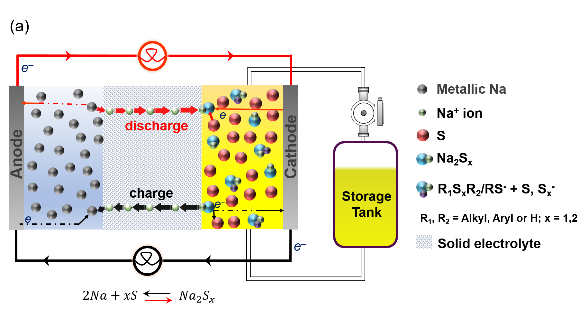
The semi-flow Intermediate Temperature (IT) Metal-Sulfur (M-Na, Li, K) battery integrates flow configuration at the cathode to improve energy density. The redox reaction of dissolved disulfides in the catholyte boosts the specific capacity of the system. The chemical interaction between S₈/Sₓ²⁻ and diorganyl disulfides (e.g., diphenyl disulfide, diethyl disulfide) or thiolate ion stabilizers (e.g., thiophenol, 2,5-dimercapto-1,3,4-thiadiazole) enhances sulfur utilization, suppresses precipitation, and improves both chemical reactivity and cycling stability.
More

The phd students’ work
